Our company has experienced experts and scientists dedicated to providing safe pharmacological testing services in large and small animals to explore the effects of drugs on the central nervous system, respiratory system and cardiovascular system for the evaluation and prediction of possible adverse effects of new drugs in human clinical trials.
Service Overview
Safety pharmacology is concerned with how medications affect the respiratory, cardiovascular, and central nervous systems when they are used within or over the therapeutic range. Observations on the urinary system, autonomic nervous system, digestive system, and other organs and tissues should also be made in follow-up and/or additional safety pharmacology investigations that may be carried out as necessary.
In vitro and in vivo safety pharmacology has become an important part of preclinical safety evaluation of new drugs. Our company provides the best safety pharmacology services at the highest quality and lowest cost.
The objectives of our services include:
- Identification of adverse pharmacological effects of drugs relevant to human safety.
- Evaluation of adverse drug reactions and/or pathophysiological effects observed in toxicological and/or clinical studies; Study of the mechanisms of observed and/or postulated adverse drug reactions.
Research Capabilities
- We can perform cardiovascular (blood pressure) and metabolic (glucose levels) assessments by continuous telemetry monitoring methods to provide high resolution data and enhance safety analysis.
- In rodents, we can monitor blood pressure by electromagnetic telemetry. Blood pressure measurements can be continued for several months if desired. Experimental readings include mean arterial pressure (MAP), systolic/diastolic blood pressure, and heart rate.
- We use DSI pressure monitoring technology to continuously monitor glucose levels in rodent and non-human primate models, thereby continuously monitoring biochemical and physiological parameters after drug administration. It has been used in a rat model and is being finalized in a mouse model. The maximum monitoring time of the telemetry device is approximately 4 weeks, which is mainly influenced by the probe lifetime (determined by the amount of glucose oxidase that can be loaded on the needle).
- In the non-human primate model, we continuously monitor blood glucose and record it remotely via a small repeater. We can monitor blood glucose wirelessly and record continuously for more than 6 months to provide high quality continuous data.
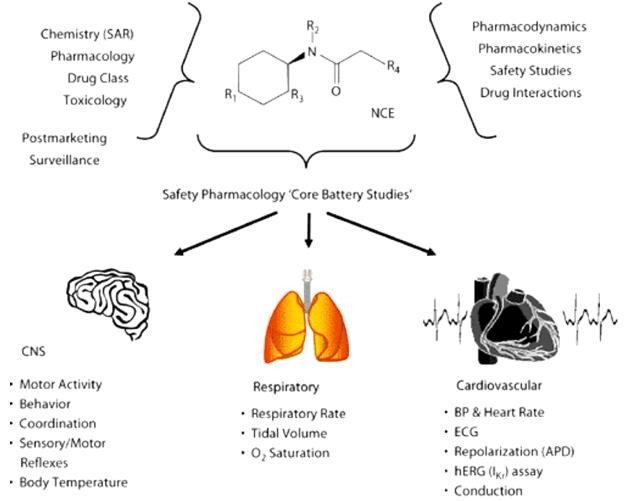 Fig 1. An overview of the multidisciplinary integration required to evaluate the safety profile of a new chemical entity (NCE) in Safety Pharmacology. (Pugsley M. K, et al. 2008)
Fig 1. An overview of the multidisciplinary integration required to evaluate the safety profile of a new chemical entity (NCE) in Safety Pharmacology. (Pugsley M. K, et al. 2008)
Overall Solution
Our company's comprehensive toxicology platform includes safety pharmacology studies to detect potential side effects of our customers' new drugs. We can provide independent safety study experiments or incorporate them into our overall toxicity analysis.
Our company's comprehensive safety pharmacology evaluation program for rodents and non-human primates is as follows.
| Systems | Non-Human Primates | Rodent |
|---|
| Cardiovascular | We monitor blood pressure (BP), heart rate (HR), electrocardiogram (ECG), and heart function through an echocardiograph. | Blood pressure and heart rate monitoring including continuous telemetry. |
| Metabolism | Long-term continuous monitoring of blood or interstitial glucose through telemetry equipment. | Long-term monitoring of glucose biomarker experiments with telemetry equipment (standard ELISA and MSD). |
| Kidney/Urine | Kidney structure, blood chemistry, protein analysis | Kidney structure, blood chemistry, kidney injury biomarker panel (MSD). |
| Liver | Fatty liver ultrasound analysis (for long-term experiments). | - |
For more information, please feel free to contact us.
Reference
- Pugsley M. K, et al. (2008). "Principles of Safety Pharmacology." Safety Pharmacology. 154(7): 1382-1399.
Related Services
It should be noted that our service is only used for research, not for clinical use.

 Fig 1. An overview of the multidisciplinary integration required to evaluate the safety profile of a new chemical entity (NCE) in Safety Pharmacology. (Pugsley M. K, et al. 2008)
Fig 1. An overview of the multidisciplinary integration required to evaluate the safety profile of a new chemical entity (NCE) in Safety Pharmacology. (Pugsley M. K, et al. 2008)